Prepackaged Food Regulatory Compliance Service in China
Background
In recent years, a large number of imported foods were destroyed or returned by Chinese CIQs due to illegal food raw materials, unqualified product quality, incompliant Chinese label and etc. The rejection has brought unnecessary loss to import-export enterprises and the company credibility was badly influenced. On 24th April 2015, the strictest food safety law ever was published to put forwards more rigorous requirements on food production, operation, inspection, supervision, punishment and etc. Related enterprises should be ready to meet the new high requirements.
What is prepackaged food?
Prepackaged food refers to the foods prepackaged or made up in advance with a measured quantity in a container and have unified labeling of quality or volume within certain range.
Related laws, regulations and national standards(Click regulation names for download)
In recent years, a large number of imported foods were destroyed or returned by Chinese CIQs due to illegal food raw materials, unqualified product quality, incompliant Chinese label and etc. The rejection has brought unnecessary loss to import-export enterprises and the company credibility was badly influenced. On 24th April 2015, the strictest food safety law ever was published to put forwards more rigorous requirements on food production, operation, inspection, supervision, punishment and etc. Related enterprises should be ready to meet the new high requirements.
What is prepackaged food?
Prepackaged food refers to the foods prepackaged or made up in advance with a measured quantity in a container and have unified labeling of quality or volume within certain range.
Related laws, regulations and national standards(Click regulation names for download)
Essential elements of qualified imported prepackaged food
- Eligible food raw materials
- Qualified product quality
- Compliant Chinese label

CIRS is experienced in food regulatory compliance service for several years, and there are professional consultants and engineers in our team. We provide professional and comprehensive food regulatory compliance service to you.
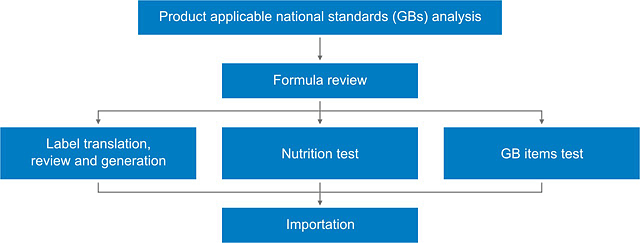
- Prepackaged food applicable Chinese national standards (GBs) analysis
- Prepackaged food formula (raw materials and food additives) review
- Prepackaged food Chinese label translation, review or generation
- Prepackaged food nutrition test
- Prepackaged food GB test
- Regulation updates monitoring
- Regulation training
- Importers, exporters and overseas manufacturers filing
- Other customized services
| S.N. | Items | Services |
| 1 | Product national standards (GBs) analysis | Evaluate the product category and find an appropriate Chinese national standards (GBs) based on the formula, production process and etc. |
| 2 | Formula (raw materials and food additives) review |
I. Analyze if all food raw materials and food additives are allowed to be used in this product. II. Analyze if the dosages of all ingredients are compliant. |
| 3 |
Chinese label translation, review or generation |
1. Label translation Translate the original information to Chinese information. 2. Label review I. Check if the Chinese label provided by client is compliant in China according to GB 7718-2011, GB 28050-2011 and product standards. II. Provide modification and risk-aversion suggestions. 3. Label generation I. Translate necessary information in the original label to Chinese. II. Generate Chinese label according to GB 7718-2011, GB 28050-2011 and product standards, such as add mandatory content or remove risky content. III. Check the label to make sure it is compliant and provide risk-aversion suggestions. |
| 4 | Nutrition test |
I. Test on protein, fat, carbohydrate, sodium and energy (4+1). II. Other nutrition test. Such as if you add nutrition enhancer into the product or there is nutritional content claim on the label, you shall test it. III. Nutrition test reports should be provided to CIQ during inspection declaration. |
| 5 | GB test |
I. GB test in a qualified lab is necessary to guarantee the product quality and avoid unnecessary rejection. II. Conduct GB test in China can help to avoid method deviation. III. For dairy product, GB testing reports should be provided to CIQ during inspection declaration. IV. CIRS will help to analyze the test items according to related product national standards, and arrange the test in qualified collaboratory in China. |
| 6 |
Regulation updates monitoring |
To help clients recognize the changes of regulations and make response in time, CIRS will deliver monthly regulation update report with the key point and influences of updated laws, regulations, national standards, and official notices to you, and corresponding suggestions from CIRS, CFDA, NHFPC, CIQs will be attached. |
| 7 | Regulation training | Customized food regulation training will be provided to help you has an overall understanding of focused legislations. |
| 8 | Importers, exporters and overseas manufacturers filing | Collate required documents and apply for filing under local CIQs (domestic consignee) or AQSIQ (overseas exporter and manufacturer). |
| 9 | Others | Ad-hoc consulting service and etc. |
Contact us
Ms. Alice Yang, Food Safety and Regulatory Affairs Department, CIRS China
11F Dongguan Building, 288 Qiuyi Road, Binjiang District, Hangzhou, China, 310020
Tel: +86 571 8971 6579 | Fax: +86 571 8720 6533
Email: Alice.Yang@cirs-group.com






